Hotline
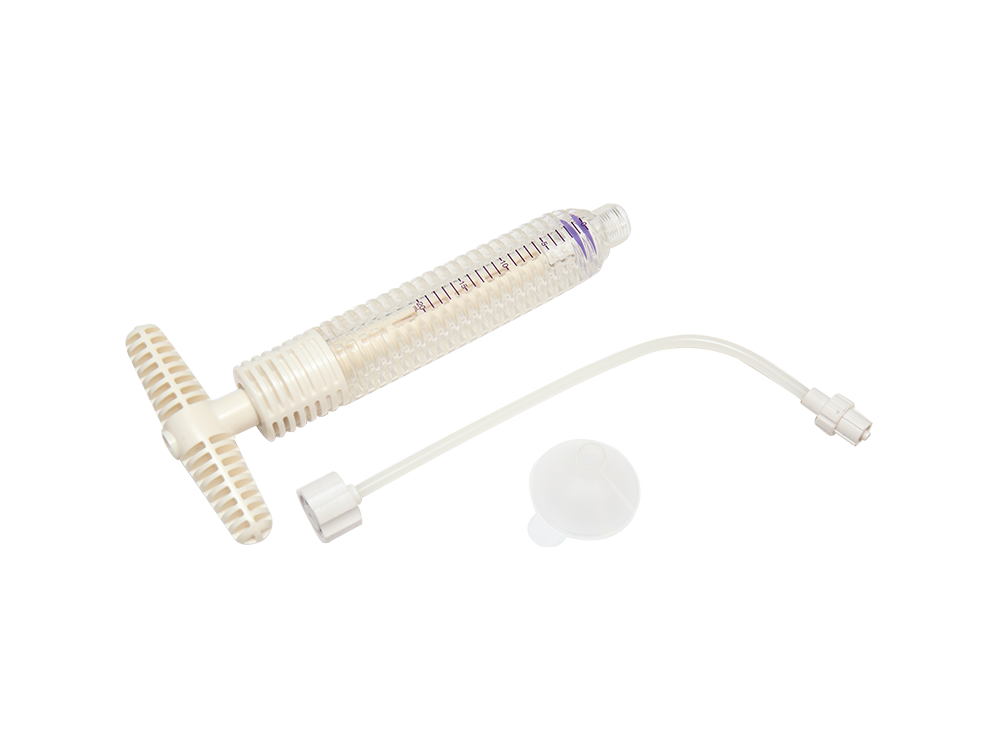
Disposable screw pusher
Disposable screw pusher
Retail price
0.0
元
Market price
0.0
元
Number of views:
1000
Product serial number
Category
Vertebroplasty products
Quantity
-
+
Stock:
1
Other Products
Vertebral body forming products
Kyphoplasty is performed under the image-enhanced assembly monitoring, percutaneously puncturing the compressed vertebral body, establishing a working channel, sending a special balloon about 15 mm in size to the center of the vertebral body, and then injecting the angiography into the balloon Agent to expand the balloon, gradually prop up the collapsed vertebral body, and partially or completely restore the height of the vertebral body. Finally, the balloon is taken out and the bone cement is filled into the "cavity" in the center of the vertebral body through the working channel. After surgery, you can quickly relieve pain; restore vertebral body height; correct kyphosis; reduce and stabilize fractures; improve patient quality of life.


Unilateral dual-channel endoscopic spinal minimally invasive
Unilateral dual-channel endoscopic technique is suitable for degenerative lesions of cervical, thoracic and lumbar spine. Unlike the single channel of the intervertebral foramen, this technique usually establishes two channels, one is the observation channel and the other is the instrument operation channel. The observation channel generally uses 0° or 30° dual-channel endoscopes. The operating channel should use dual-channel special instruments, such as dual-channel scraping spoons, dual-channel magic drills (dual-channel endoscope grinding heads), dual-channel endoscope planers, Double-channel endoscopic plasma cutter head, double-channel lamina bone pliers, double-channel nucleus pulposus forceps, double-channel nerve retractor, etc. The dual-channel technology has a special equipment package, and the unilateral dual-channel endoscopic technology can also complete the fusion operation under the microscope.


Skull bone cement
Polymethyl methacrylate (PMMA) is a polymer obtained from acrylic acid and its ester polymers and is collectively referred to as acrylic resin.



Blon public account

Mobile Website
Blon Medical Technologies
Address: 3230 Fallow Field Dr.Diamond Bar, CA 91765, USA
E-mail: info@blonmedicaltech.com
Tel:909 655-8119
Cooperation Hotline
+86-0519-86470196
Address: No. 525 Changwu Road, Wujin High-tech Development Zone, Changzhou City, Jiangsu Province
Tel:+86-0519-86470196 / 13801500434
E-mail:info@blonmedical.com
Fax:+86-0519-86488930
Copyright © 2019 Changzhou Blon Minimally Invasive Medical Devices Technology Co. Ltd. 苏ICP备19073665号-1